

| (40) |
T is the temperature of the emitters in K, and M the atomic
weight in atomic mass units (amu).
Resonance broadening (self-broadening) occurs only between identical
species and is confined to lines with the upper or lower level having an
electric dipole transition (resonance line) to the ground state. The FWHM may
be estimated as
|
|
(41) |
where λ is the wavelength of the observed line. fr and λr are the oscillator strength and wavelength of the resonance line; gk and gi are the statistical weights of its upper and lower levels. Ni is the ground state number density.
For the 1s2p 1P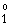 - 1s3d
1D2 transition in He I
[λ = 6678.15 �;
λr (1s2
1S0 - 1s2p
1P
- 1s3d
1D2 transition in He I
[λ = 6678.15 �;
λr (1s2
1S0 - 1s2p
1P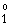 )
= 584.334 �; gi = 1;
gk = 3;
fr = 0.2762] at
Ni = 1 � 1018 cm-3:
ΔλR1/2 = 0.036 �.
)
= 584.334 �; gi = 1;
gk = 3;
fr = 0.2762] at
Ni = 1 � 1018 cm-3:
ΔλR1/2 = 0.036 �.
Van der Waals broadening arises from the dipole interaction of an
excited atom with the induced dipole of a ground state atom. (In the case of
foreign gas broadening, both the perturber and the radiator may be in their
respective ground states.) An approximate formula for the FWHM, strictly
applicable to hydrogen and similar atomic structures only, is
|
|
(42) |
where � is the atom-perturber reduced mass in units of u, N the perturber density, and C6 the interaction constant. C6 may be roughly estimated as follows: C6 = Ck - Ci, with Ci (k) = (9.8 � 1010) (αd R2i (k) αd in cm3, R2 in a02). Mean atomic polarizability αd ≈ (6.7 � 10-25) (3IH/4E*;)2 cm3, where IH is the ionization energy of hydrogen and E*; the energy of the first excited level of the perturber atom. R2i (k) ≈ 2.5 [IH/(I-Ei (k))]2, where I is the ionization energy of the radiator. Van der Waals broadened lines are red shifted by about one-third the size of the FWHM.
For the 1s2p 1P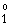 - 1s3d 1D2
transition in He I, and with He as perturber:
λ = 6678.15 �;
I = 198 311 cm-1;
E* = Ei = 171 135 cm-1;
Ek = 186 105 cm-1;
� = 2. At T = 15 000 K and
N = 1 � 1018 cm-3:
ΔλW1/2 = 0.044 �.
- 1s3d 1D2
transition in He I, and with He as perturber:
λ = 6678.15 �;
I = 198 311 cm-1;
E* = Ei = 171 135 cm-1;
Ek = 186 105 cm-1;
� = 2. At T = 15 000 K and
N = 1 � 1018 cm-3:
ΔλW1/2 = 0.044 �.
Stark broadening due to charged perturbers, i.e., ions and
electrons, usually dominates resonance and van der Waals broadening in
discharges and plasmas. The FWHM for hydrogen lines is
| (43) |
| Ne (cm-3) | ||||
|---|---|---|---|---|
| T(K) | 1015 | 1016 | 1017 | 1018 |
| 5 000 | 0.0787 | 0.0808 | 0.0765 | ... |
| 10 000 | 0.0803 | 0.0840 | 0.0851 | 0.0781 |
| 20 000 | 0.0815 | 0.0860 | 0.0902 | 0.0896 |
| 30 000 | 0.0814 | 0.0860 | 0.0919 | 0.0946 |